Crystal Mounts and Loops
Simplify crystal harvesting, and obtain the best possible data from your crystals, using the most advanced lithographic cryo-loops available.
Composed of X-ray transparent material, crystals can be mounted directly on the loops, reducing excess fluid, preventing damage, and making getting the crystals out of the drop easier.
Learn More Below Improve Crystal Harvesting with Humidification – Learn More Selection Guide – Choose The Best LoopProduct Information
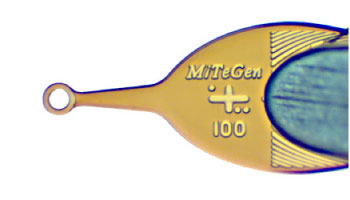
MicroMounts™ are the world's highest performance tools for retrieving and mounting protein crystals, virus crystals, and small molecule/inorganic crystals. They can be also used for handling small fragile samples of all sorts, including biological, archaeological and mineralogical samples. MicroMounts™ are compatible with all standard X-ray hardware, and can be inserted in 0.7 mm mechanical pencils or micromanipulators for easy handling.
Micromounts™ consist of thin polymer tips attached to beveled non-magnetic 0.025" (0.64 mm) diameter stainless steel rods. Their patented design provides an excellent combination of X-ray and optical transparency, mechanical rigidity and flexibility, and precision dimensions.
The polymer is unoriented, so the background scatter does not show the sharp peaks of competing mounts, and has the lowest possible birefringence.
Curving the tip by wrapping its base around the rod makes it rigid, and provides a scoop-like action in retrieving samples. The tip can also flex and bend flat against e.g., a well or glass slide, to dislodge and then slip under a sample.
A wicking aperture draws excess liquid away from the sample, reducing background scatter and allowing faster sample cooling for cryocrystallography.
Since 2008, MicroMounts™ (and all of our other products) have been made using the most mechanically robust unoriented polymer available. With proper care - especially during cleaning - each MicroMount™ can be used to harvest and measure dozens of crystals.
Extremely easy to use, novices can be mounting samples in minutes. MicroMounts™ make your crystallography pipeline more efficient by maximizing the odds of successful mounting, cooling and data collection.
You may also like…
-
Universal Puck (Uni-Puck) Starter Kits
To get you up–and–running sending samples to the beamline in easy-to-order bundles. Full Description -
Reusable Goniometer Base Styles
Quickly assemble MiTeGen Mounts, Loops, and Meshes into bases for crystal harvesting, without using glue or... -
In Situ-1 Crystallization Plate
The In Situ‐1™ plate enables crystallography researchers to easily grow, transport and screen crystals of...